Join Dr. Tsai and Dr. Kumar on a journey into novel treatment avenues for Fragile X syndrome. Learn how activating mGluR7 could be a game-changer, opening up uncharted therapeutic territory.
Read moreHas Results
Fragile X Clinical Trial of New PDE4D Inhibitor from Tetra

With a $200,043 grant from FRAXA Research Foundation, Dr. Elizabeth Berry-Kravis completed a successful Phase 2 clinical trial of a PDE4 inhibitor for adult men with Fragile X syndrome. This trial treated 30 males, 18-45 years of age with a new PDE4D allosteric inhibitor from Tetra Discovery Partners using a crossover design, so that everyone got active drug for part of the time and placebo for part of the time.
Read moreRecruiting: Clinical Study of Non-Invasive EEG for Children Ages 2-7

Dr. Carol Wilkinson, MD PhD, and Dr. Charles Nelson, PhD, at Boston Children’s Hospital are recruiting children ages 2-7 years with Fragile X syndrome to participate in a study of brain differences using non-invasive EEG.
Read moreLovamix: Clinical Trial of Combined Treatment of Minocycline and Lovastatin in Fragile X Syndrome

With a $66,714 grant from the FRAXA Research Foundation awarded over 2015-2017, Dr. Francois Corbin at the Universite of Sherbrooke will test the safety and synergistic effects of lovastatin and minocycline in patients with Fragile X syndrome.
Read morePositive Results Reported in Phase II Fragile X Clinical Trial of PDE4D Inhibitor Zatolmilast from Tetra Therapeutics

Today, Tetra Therapeutics announces the first unequivocally positive phase 2 clinical trial in Fragile X syndrome, press release below. The results do not depend on carving out a subset of patients or post hoc analysis.
Read moreClinical Trial of Ganaxolone in Patients with Fragile X Syndrome

With a $90,000 grant from FRAXA Research Foundation funded during 2014-2015, Dr. Frank Kooy and colleagues at the University of Antwerp are conducting a double blind crossover trial of ganaxolone in patients with Fragile X syndrome. Results of this study were mixed (see Marinus: Results from Phase 2 Exploratory Clinical Study Support Continued Development of Ganaxolone in Fragile X Syndrome.)
Read moreNeuren’s Tofinetide Successful in Phase 2 Clinical Trial in Fragile X
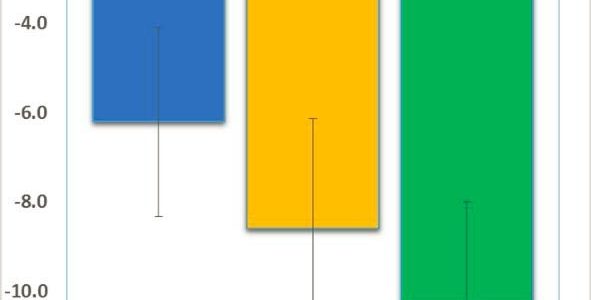
We are pleased to share great news adapted from Neuren’s press release: Neuren’s phase 2 trial has successfully established proof of concept and provides a strong rationale for Neuren to move forward with developing trofinetide for Fragile X syndrome. In this initial small trial with a relatively short treatment period, trofinetide was very well tolerated, with the high dose (70 mg/kg twice daily) demonstrating a consistent pattern of clinical improvement, observed in both clinician and caregiver assessments.
Read morePhase 1 Clinical Trial of Mega Green Tea Extract in Fragile X Syndrome

With a $124,000 grant from the FRAXA Research Foundation from 2012-2014, Dr. Mara Dierssen and Dr. Rafael de la Torre conducted preclinical studies in Fragile X knockout mice and a clinical trial in Fragile X patients using Mega Green Tea Extract, which contains 45% by weight epigallocatechin gallate (EGCG).
Read morePilot Clinical Trial of Lithium in Fragile X Shows Promising Results

With a $65,000 grant from FRAXA Research Foundation in 2005, Dr. Berry-Kravis at the Rush University Medical Center conducted a pilot clinical trial of lithium in 15 patients with Fragile X syndrome. Results published.
Read moreEncouraging Results from First Trial of Minocycline in Fragile X

With a $40,000 grant from FRAXA, Dr. Carlo Paribello and his team at the Surrey Place Centre Fragile X clinic in Toronto, Ontario, ran an open label trial to see if minocycline can improve learning and reduce anxiety and behavioral problems in people with Fragile X. Twenty participants between the ages of 13 and 35 years took minocycline for two months.
Read moreClinical Trial of Aripiprazol in Fragile X Syndrome

With a FRAXA Research Foundation grant of $30,000 in 2006, Dr. Erickson conducted a pilot clinical trial of an available medicine, aripiprazole (brand-name Abilify). This was an open-label 12-week trial in 12 people ages 6–25 years with Fragile X. Results were promising, and published: 10 of the 12 participants showed behavioral improvements.
Read moreMelatonin Clinical Trial in Fragile X

With a $60,000 grant from FRAXA Research Foundation from 1998-1999, Dr. Randi Hagerman and her team at the University of California studied the effects of different compounds on individuals with Fragile X syndrome, focusing specifically on melatonin. Results published.
Read more
