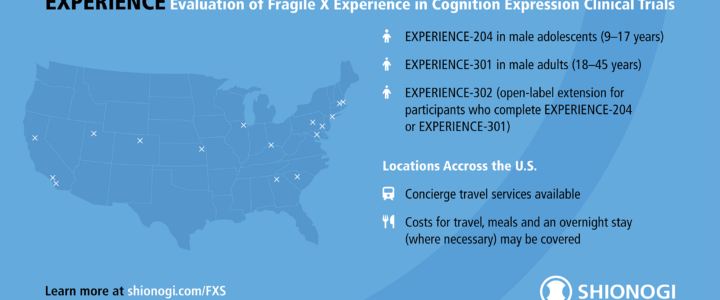
Shionogi’s EXPERIENCE clinical trials for Fragile X syndrome are nearing completion. Enrollment for the adult trial (EXPERIENCE-301) is now closed, while the adolescent trial (EXPERIENCE-204) is in its final phase. Learn more about the study, FRAXA’s role, and the open-label extension.
Read moreFDA
NPR Spotlights Zatolmilast: A Potential Breakthrough for Fragile X Syndrome

NPR spotlights zatolmilast, a promising drug offering new hope for individuals with Fragile X syndrome. Families report life-changing improvements in anxiety, communication, and independence.
Read moreBK Channel Openers: A New Drug for Fragile X Is Ready for Clinical Trials
Discover the promising new BK channel opener, SPG601, now entering clinical trials for Fragile X syndrome. Learn about its potential to restore synaptic function and address core symptoms.
Read moreASOs and Fragile X: Addressing the Most Asked Questions

Explore the potential of ASOs in treating Fragile X syndrome & FXTAS. Dive into a comprehensive Q&A addressing key questions and breakthrough findings.
Read morePioneering Community-Based Drug Development for Fragile X Syndrome

Discover how FRAXA leverages Community-Based Drug Development to create impactful therapies for Fragile X syndrome. Join us as we reshape the future of rare disease treatment.
Read moreAllos Pharma Advances Phase 3 Clinical Trial Design for Potential Fragile X Syndrome Treatment, Arbaclofen

Discover Allos Pharma’s advancements in a pivotal Phase 3 trial for Fragile X syndrome treatment, Arbaclofen. Learn how their FDA-informed trial design might finally bring hope to the Fragile X community.
Read moreSRC Family Kinase Inhibitor as a Potential Treatment for Fragile X Syndrome

Discover the Smith lab’s new approach to treating Fragile X syndrome using Saracatinib, originally a cancer drug. Learn how this $100,000 FRAXA research grant-funded study opens up hopeful avenues.
Read moreFragile X Clinical Trial of New PDE4D Inhibitor from Tetra

With a $200,043 grant from FRAXA Research Foundation, Dr. Elizabeth Berry-Kravis completed a successful Phase 2 clinical trial of a PDE4 inhibitor for adult men with Fragile X syndrome. This trial treated 30 males, 18-45 years of age with a new PDE4D allosteric inhibitor from Tetra Discovery Partners using a crossover design, so that everyone got active drug for part of the time and placebo for part of the time.
Read moreRepurposing FDA-Approved Drugs to Treat Major Depressive Disorder in Fragile X Syndrome

Did you know that depression is more common in those with autism and/or Fragile X? Even more disturbing is the discovery that current treatments for depression do not work in Fragile X mice. With this grant, the team will work to develop a rapid screening tool to identify FDA-approved drugs which can treat depression in people with Fragile X syndrome.
Read moreNew Fragile X Clinical Trial Announced by Healx

Healx’s AI-driven approach makes finding the right combination therapies more efficient, cost-effective, and rapidly ready for testing at FRAXA-DVI. It was this process that has brought Healx to its recent announcement sharing that it has received Investigational New Drug (IND) approval from the US Food and Drug Administration (FDA) for the Phase 2a clinical study of HLX-0201 (sulindac, an FDA-approved drug).
Read more2021 Fragile X Research Grants Funded by FRAXA Research Foundation

Each year, FRAXA funds a diverse portfolio of research. Our FRAXA Fellowships are seed funding for the future, the feedstock for the Fragile X treatment development pathway. While we are looking to promote as many promising new approaches as possible, prominent themes emerge each year, as scientists around the world tackle previously neglected areas.
Read moreSynaptogenix Announced Intention to Launch a Fragile X Clinical Trial with Bryostatin

One of the most exciting kinds of work that FRAXA does is following the journey of an experimental new treatment until it is ready for trials in people with Fragile X. From an initial idea, through the development process, to clinical trials, FRAXA helps out all along the way. From an initial idea, through the development process, to clinical trials, FRAXA helps out all along the way. The recent announcement by Synaptogenix is a great example of how FRAXA funding and use of FRAXA-DVI can accelerate research on Fragile X.
Read morePivotal Phase 3 Trial of Zygel in Severe Fragile X Possible This Year

Zynerba Pharmaceuticals reported receiving advice from the U.S. Food and Drug Administration (FDA) on the design of an upcoming Phase 3 clinical trial meant to confirm previous trial findings supporting Zygel as a cannabidiol treatment in a specific subset of Fragile X syndrome patients. The new trial, called RECONNECT, is expected to launch before October, and will mainly enroll children and adolescents with a complete (100%) methylation of FMR1, the gene mutated in Fragile X.
Read moreCOVID-19 Vaccines Pose Little Risk to Rare Disease Patients, FDA, CDC Say
The two COVID-19 vaccines that recently received emergency approval from the US and other worldwide regulatory agencies are expected to pose little risk to the rare disease community, including to patients with compromised immune systems or those participating in gene therapy studies.
Read moreBryostatin-1 in Long-term Use Seen to Arrest Fragile X Symptoms in Mouse Model
Long-term, but not short-term, treatment with bryostatin-1 — Neurotrope’s lead investigational therapy — arrested such behavioral and cognitive symptoms as hyperactivity, difficulties with daily life activities, and learning and memory deficits in a mouse model of Fragile X syndrome.
Read morePositive Results Reported in Phase II Fragile X Clinical Trial of PDE4D Inhibitor Zatolmilast from Tetra Therapeutics

Today, Tetra Therapeutics announces the first unequivocally positive phase 2 clinical trial in Fragile X syndrome, press release below. The results do not depend on carving out a subset of patients or post hoc analysis.
Read moreZygel May Improve Behavior in Children With Severe Fragile X, Trial Data Suggest

Zynerba presented clinical trial results for Zygel at a recent neurology conference. Zygel, an experimental cannabidiol (CBD) gel, may reduce behavioral abnormalities in children with Fragile X syndrome who have more severe disease.
Read moreAripiprazole (Abilify) in the Treatment of People with Fragile X: An Anecdotal Account

The aim of this article is to discuss the use of Abilify (generic name: aripiprazole) as a treatment for people with Fragile X syndrome (FXS). As an “off-label” prescription, Abilify targets behaviors such as irritability, aggression, self-injury and severe tantrums.
Read moreBrain Organoids and Therapeutic Development for Fragile X and Other Rare Diseases

This is the first in a series of webinars focused on current topics in Fragile X research. In this webinar we hear from Alysson R. Muotri, PhD, Professor at University of California San Diego Stem Cell Programand Fabio C. Tucci, PhD, Chief Operating Officer and co-founder at Epigen Biosciences, Inc.
Read moreCompanies Move to Advance Potential Cognitive Treatment for Fragile X

Tetra Therapeutics and Shionogi announced plans to expand their partnership supporting BPN14770, a treatment candidate for disorders marked by cognitive and memory deficits, including Fragile X syndrome and Alzheimer’s disease. The agreement builds on an earlier collaboration between the two companies, and aims to further accelerate BPN14770’s development and potential marketing. It is currently in clinical testing in both Fragile X and Alzheimer’s patients.
Read moreResults Reported: Using EEG Responses to Sound for Fragile X Drug Discovery

Jonathan Lovelace, a FRAXA funded Postdoc at UC Riverside, has made some exciting EEG findings over the past few years studying auditory hypersensitivity in mice and therapeutic drug treatments. A big obstacle in FXS research has been establishing reliable, unbiased, and translation relevant biomarkers that can be used to determine the effectiveness of therapies. One of the most important discoveries they have made is the striking similarity in EEG biomarkers between mice and humans.
Read moreScreening 2,320 FDA-Approved Drugs for Potential Treatment of Fragile X

FRAXA Research Foundation has awarded a $90,000 grant to Principal Investigator Dr. Sean McBride and Postdoctoral Fellow Dr. Karen Joyce, at Rowan University, to screen all 2,320 FDA-approved drugs on both mouse and fly models of Fragile X syndrome. Those drugs which show promise will be tested in more detail for potential to treat Fragile X in humans.
Read moreTetra Announces $40M to Advance BPN14770 for FXS and Alzheimer’s Disease

Tetra Discovery Partners has signed a multi-part deal that could bring it up to $160 million, plus royalties, from Shionogi & Co, Ltd, a Japanese major research-driven pharmaceutical company. Tetra currently is conducting an investigational Phase 2 study of BPN14770 in adults with Fragile X Syndrome, an indication for which BPN14770 has received Orphan Drug Designation from the US Food and Drug Administration. This clinical trial was made possible by early work with the FRAXA-DVI and over $200,000 from FRAXA.
Read moreTetra Discovery Partners Initiates Phase 2 Trial of BPN14770 in Fragile X Syndrome

This 2-Period Crossover Study of BPN14770 is accepting adults males with Fragile X syndrome at Rush University Medical Center in Chicago. Principal Investigator of the study is Elizabeth Berry-Kravis, MD, PhD.
A selective inhibitor of the phosphodiesterase type-4D (PDE4D), BPN14770 has shown the ability to improve the quality of connections between neurons and to improve multiple behavioral outcomes in the Fragile X mouse model.
Takeaways from Fragile X Advocacy Day

In the first week of March I attended my first Fragile X Advocacy Day to meet with many of the Massachusetts delegation to Congress. While this was my first time advocating for Fragile X research, I’ve been a longtime lung cancer research advocate and have met with many of the same representatives in the past. It was a pleasure to meet with many of the families as my participation in Advocacy Day was in the spirit of “we are all in this together”.
Read more